-
 Ohmic conductor
Ohmic conductor
-
 Dorsum
Dorsum
-
 Undulated green slate
Undulated green slate
-
 Immunomodulator
Immunomodulator
-
 SPOT 5
SPOT 5
-
 Chondrite
Chondrite
-
 Nuclear reaction
Nuclear reaction
-
 Cytokinesis
Cytokinesis
-
 Heliocentric
Heliocentric
-
 Ikonos
Ikonos
-
 Acidophile
Acidophile
-
 WPA
WPA
-
 Cotylopus acutipinnis
Cotylopus acutipinnis
-
 Catabolism
Catabolism
-
 Spirit
Spirit
-
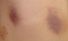 Ecchymosis
Ecchymosis
-
 SETI
SETI
-
 Proterozoic era
Proterozoic era
-
 Amylase
Amylase
-
 Adjuvant
Adjuvant
-
 NexGuard
NexGuard
-
 Sedge
Sedge
-
 Gas planet
Gas planet
-
 Sclerotin
Sclerotin
-
 Food additive
Food additive
-
 Abnormality
Abnormality
-
 Systematic
Systematic
-
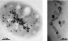 Magnetotactic bacterium
Magnetotactic bacterium
-
 Quantum entanglement
Quantum entanglement
-
 Tau
Tau
Photochemical pollution
Photochemical pollution, also called photo-oxidant pollution, is caused by the transformation of primary pollutants, chemical precursors, into oxidant forms when exposed to sunlight.
After exposure to ultraviolet rays, photochemical reactions transform nitrogen oxides (NOx) and volatile organic compounds (VOC) into tropospheric ozone (O3) and other oxidant compounds (aldehydes, hydrogen peroxide, etc.)
Photochemical pollution is therefore responsible for ozone peaks and their harmful effects on human populations as well as plants. Moreover, oxidant forms also cause the acidification of soil and water.
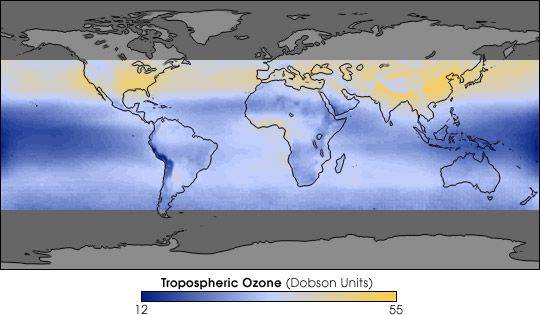 Cartography of the tropospheric ozone, a source of photochemical pollution, in August from 1979 to 2000. The blue areas indicate a decrease in levels, the yellow areas indicate an increase in levels. © Jack Fishman / Nasa Langley Research Center
Cartography of the tropospheric ozone, a source of photochemical pollution, in August from 1979 to 2000. The blue areas indicate a decrease in levels, the yellow areas indicate an increase in levels. © Jack Fishman / Nasa Langley Research Center
Latest
Fill out my online form.



