-
 Ramjet engine
Ramjet engine
-
 PSA
PSA
-
 VPN
VPN
-
 Fayalite
Fayalite
-
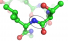 Protease
Protease
-
 Hornfel
Hornfel
-
 DMT
DMT
-
 Rhesus factor
Rhesus factor
-
 String theory
String theory
-
 Peering
Peering
-
 Thermodynamics
Thermodynamics
-
 Extinction
Extinction
-
 Ocular circle
Ocular circle
-
 Menopause
Menopause
-
 Scambaiting
Scambaiting
-
 NAT
NAT
-
 Light pen
Light pen
-
 Radioactivity
Radioactivity
-
 Western Palearctic ecozone
Western Palearctic ecozone
-
 Translation
Translation
-
 Artemisin
Artemisin
-
 Ecosystem
Ecosystem
-
 Bus
Bus
-
 Parsnip
Parsnip
-
 Carbonado
Carbonado
-
 Position isomerism
Position isomerism
-
 Covolume
Covolume
-
 Pigmentation
Pigmentation
-
 Reach
Reach
-
 Heuristics
Heuristics
Valency
The valency (or valence) of an atom determines the number of bonds that it can form with other atoms. For example, a valency of 1 indicates that an atom can only form a bond with one other atom; a valency of 3 that it can form bonds with three other atoms, etc. We say that an atom is monovalent, divalent, trivalent, tetravalent, pentavalent, hexavalent etc.
The noble gases (helium, neon, argon, krypton, xenon, radon),have a valency of 0 and therefore only occur naturally in the monatomic form.
Latest
Fill out my online form.



