-
 Prompt
Prompt
-
 Casimir effect
Casimir effect
-
 Guttation
Guttation
-
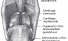 Larynx
Larynx
-
 Garden strawberry
Garden strawberry
-
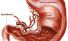 Stomach
Stomach
-
 Restriction enzymes
Restriction enzymes
-
 Tourette's syndrome
Tourette's syndrome
-
 Glycaemia
Glycaemia
-
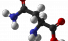 Asparagine
Asparagine
-
 Tempel 1 comet
Tempel 1 comet
-
 Splicing
Splicing
-
 Thermostable
Thermostable
-
 Adrenaline
Adrenaline
-
 Exudate
Exudate
-
 Crinoids
Crinoids
-
 Catheter
Catheter
-
 Pyrophyllite
Pyrophyllite
-
 IPTV
IPTV
-
 Chloracne
Chloracne
-
 Costal
Costal
-
 Mammal
Mammal
-
 Gliding.
Gliding.
-
 High definition television
High definition television
-
 Gaviiformes
Gaviiformes
-
 Volcano
Volcano
-
 Complete metamorphosis
Complete metamorphosis
-
 Stratiform
Stratiform
-
 Rhesus macaque
Rhesus macaque
-
 Ohm
Ohm
Thermal conductivity
In physics, thermal conductivity is the quantity that measures the ability of a body to conduct heat. It is the quantity of heat transferred per unit surface area and per unit of time under the action of a difference in temperature between the two ends of a sample of this body and hence of a temperature gradient.
It depends mainly on the nature of the material and is usually found to have a direct relationship with the electrical conductivity of a body. This property, which may appear surprising at first, was simply explained by the fact that it is the free electrons in a body that are usually associated with heat transfer.
There are exceptions though, such as diamond which clearly shows that another possible heat conduction process is at work. This time it is the quantification of the oscillation movements of the atoms around their equilibrium position that leads to the existence of a new type of energy quantum called phonons. It is these that then play a role analogous to electrons in heat conduction.
Thermal conductivity is involved in the formulation of Fourier's law for heat conduction.
Latest
Fill out my online form.



