-
 Prophage
Prophage
-
 Anti-oxidant
Anti-oxidant
-
 European corn borer
European corn borer
-
 Intumescent
Intumescent
-
 Menstrual cycle
Menstrual cycle
-
 Black poplar
Black poplar
-
 Gost
Gost
-
 Networking
Networking
-
 Electronic message service
Electronic message service
-
 Monogyny
Monogyny
-
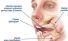 Salivary gland
Salivary gland
-
 Raman effect
Raman effect
-
 Glottis
Glottis
-
 Vanograph
Vanograph
-
 Brood
Brood
-
 Isomerism
Isomerism
-
 Specialised link
Specialised link
-
 Soma
Soma
-
 Ghost lineage
Ghost lineage
-
 EUCEB
EUCEB
-
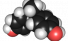 Bisphenol A
Bisphenol A
-
 Mesozone
Mesozone
-
 Neoformation
Neoformation
-
 Kuiper belt
Kuiper belt
-
 Ultrafiltration
Ultrafiltration
-
 Enzymopathy
Enzymopathy
-
 WUSB
WUSB
-
 Viking
Viking
-
 Mandible
Mandible
-
 Vitamin A
Vitamin A
Solid solution
A solid solution is a mixture formed by dissolving the atoms of an element in a metallic crystal lattice of a second element; the composition of the solid solution is homogeneous and its properties are intermediate between those of several pure constituents. (Garnets are in fact solid solutions with three mineral components).
Two cases are possible:
- either the element is incorporated in the unit cell next to or between two atoms of the principle element: this is an interstitial solid solution;
- or the incorporated element takes the place of an atom of the principle element in the crystal lattice; this is a substitutional solid solution.
Examples of solid solutions
- The plagioclases are continuous solid solutions of which the composition varies from sodic albite, (Si3AlO8)Na, to anorthite, (Si2Al2O8)Ca.
- Alkaline feldspars are a solid solution between potassic orthoclase, (Si3AlO8)K, and sodic albite, unstable at low temperature.

Plagioclase is a solid solution. © Wikipedia, Jurema Oliveira, DP
Latest
Fill out my online form.



