-
 Hyperaluminous
Hyperaluminous
-
 Constellation of Ursa Major
Constellation of Ursa Major
-
 Strong opioid analgesic
Strong opioid analgesic
-
 DRCS
DRCS
-
 Flex fuel
Flex fuel
-
 CEDRE
CEDRE
-
 M95
M95
-
 Cervical vertebra
Cervical vertebra
-
 Proper time
Proper time
-
 Amyotrophy
Amyotrophy
-
 Primaries (feathers)
Primaries (feathers)
-
 MediaFLO
MediaFLO
-
 Apomixis
Apomixis
-
 Tannification
Tannification
-
 Mercalli scale
Mercalli scale
-
 Server
Server
-
 IAM
IAM
-
 Efficacy
Efficacy
-
 EFSA
EFSA
-
 Ligo
Ligo
-
 Cystitis
Cystitis
-
 Verification
Verification
-
 Hyakutake's comet
Hyakutake's comet
-
 WEP
WEP
-
 Eyepiece
Eyepiece
-
 Neritic zone
Neritic zone
-
 Chemical graining
Chemical graining
-
 Web of Trust
Web of Trust
-
 CO2 trap
CO2 trap
-
 Remission
Remission
Hardness
The term hardness is used in various fields. It is essentially used to characterise water but is also used in mineralogy and physics.
The hardness of water
The hardness of a particular water is its concentration in calcium (Ca2+) and magnesium (Mg2+) ions. Different degrees are used in different countries; in the UK, English degrees (°e) and in the USA, ppm, also called American degrees. 1°e is equivalent to 2.80 mg of calcium ions or 1.68 mg of magnesium ions per litre. 1 ppm is equivalent to 0.4 mg of calcium ions or 0.24 mg of magnesium ions per litre.
The higher the value, the harder the water.
Soft water has a low mineral content and a high capacity to dissolve minerals including heavy metals such as the lead from old water pipes.
Mineralogy
In mineralogy hardness is the resistance of a mineral to scratching. The Mohs scale classifies hardness in relation to ten test minerals (10: diamond, 9: corundum, 8: topaz, 7: quartz, etc.).
Physics
Measures the ability of a material to resist deformation.
The test methods to determine hardness are:
- the Brinell test: the penetration of a ball of diameter D under a load F into the material is measured by measuring the diameter D of the indent and calculating the area S of the spherical section; then the Brinell hardness = F/S;
- the Rockwell test: the successive penetrations (e) of a 120° conical diamond tip are measured under loads of F0 (e0), F1+F0 (e1) and F0 (e2) (persistence). e3 = e2 -e0, e = e3/0.002 is calculated and HRC (Rockwell hardness) = 100 - e ;
- the Vickers test: a pyramid-shaped diamond with a square base under a load F is applied for a given time; the diagonal of the indent (d) is measured and the surface area S of the indent is calculated, and HV (Vickers hardness) = F/S;
- the Shore test: a ball of mass m and diameter D is dropped from a height H and the rebound height h is measured. The energy of the fall is absorbed by a plastic deformation (without indent) of the material.
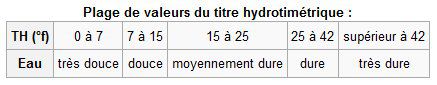
The classification of water hardness. © Wikipedia, CC by-sa
Latest
Fill out my online form.



