-
 Specific electricity
Specific electricity
-
 Mass number
Mass number
-
 Velociraptor
Velociraptor
-
 Cephalin
Cephalin
-
 BCD galaxy
BCD galaxy
-
 Compton scattering
Compton scattering
-
 Phanerozoic
Phanerozoic
-
 Stockholm Conference
Stockholm Conference
-
 Water primrose
Water primrose
-
 Tourette's syndrome
Tourette's syndrome
-
 Petrified
Petrified
-
 Haemolytic anaemia
Haemolytic anaemia
-
 Pulse
Pulse
-
 Cystoscopy
Cystoscopy
-
 Inertia
Inertia
-
 Nitrogen cycle
Nitrogen cycle
-
 Nosocomial
Nosocomial
-
 Ocean turbine
Ocean turbine
-
 Affect
Affect
-
 Neovascularisation
Neovascularisation
-
 Polymer
Polymer
-
 FAO
FAO
-
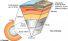 Mantle
Mantle
-
 Dyslexia
Dyslexia
-
 Polyunsaturated fatty acid
Polyunsaturated fatty acid
-
 Planking
Planking
-
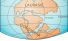 Gondwana
Gondwana
-
 In situ
In situ
-
 Condom
Condom
-
 Colloidal aggregate
Colloidal aggregate
Semi-conductor
A semi-conductor is a crystalline substance with an electrical conductivity intermediate between that of metals and that of insulators.
Unlike a metal, a semiconductor becomes an insulator at a temperature of zero kelvin (absolute zero).
Semiconductors are widely used in electronics for making components such as diodes, transistors, thyristors and integrated circuits, as well as semiconductor lasers.
Physics The intrinsic conductivity of a semiconductor crystal is related to the number of intrinsic charge carriers (electrons or holes). When a conduction electron is freed, i.e. when it goes from the valence band to the conduction band, it leaves a hole in the valence band, and this hole takes part in electrical conduction like an electron with a positive charge.
The lower the energy difference between the conduction band and the valence band (or gap), the easier it is to form an electron-hole pair.
Intrinsic semiconductors have a characteristic property: thermal agitation is sufficient to provide the energy necessary for the electron to go from the valence band to the conduction band. The resulting intrinsic conductivity increases with temperature, unlike what happens with a metallic conductor. Electromagnetic radiation can also cause an electron to go to the conduction band (photoconduction).
Extrinsic semiconduction is obtained through the introduction of impurities, by n doping (negative) or p doping (positive), and this greatly increases the conductivity of intrinsic semiconductors.
The free carriers introduce an extra energy band into the forbidden band of the semiconductor (with acceptors or donors, according to whether there is p or n doping).
Latest
Fill out my online form.



