-
 Responsivity
Responsivity
-
 Full Duplex
Full Duplex
-
 Fetus
Fetus
-
 Osteosarcoma
Osteosarcoma
-
 Carbohydrate
Carbohydrate
-
 Plate tectonics
Plate tectonics
-
 Transplant
Transplant
-
 Ulysses
Ulysses
-
 Ammonia
Ammonia
-
 Dwarf planet
Dwarf planet
-
 Scrolling
Scrolling
-
 POP
POP
-
 Rare earth minerals
Rare earth minerals
-
 Ikonos
Ikonos
-
 Nematode
Nematode
-
 Earth Summit
Earth Summit
-
 Introduced species
Introduced species
-
 SSL
SSL
-
 SDSI
SDSI
-
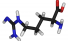 Arginine
Arginine
-
 Minor axis
Minor axis
-
 Ozone layer
Ozone layer
-
 Sexual selection
Sexual selection
-
 ATV-CC
ATV-CC
-
 Biogeography
Biogeography
-
 Numbers
Numbers
-
 Lander
Lander
-
 Gravitational wave
Gravitational wave
-
 Sex glands
Sex glands
-
 Lithification
Lithification
Optical isomerism
Two molecules or groups are optical isomers if their structures differ only by their geometry (which gives rise to different optical activities and perhaps physical properties).
The most common example is when there are asymmetric carbons in different configurations in the two molecules: cis and trans configurations around a double bond etc.
History of science Optical isomerism was studied by Pasteur, who separated the levorotatory and dextrorotatory forms of sodium ammonium tartrate (1848). Stereochemistry was founded on the work of Le Bel and van't Hoff (1875), who related the properties of the optical isomerism of certain compounds to the tetrahedral structure of carbon and the presence of asymmetric carbons.
Latest
Fill out my online form.



