-
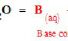 Acid
Acid
-
 Magnetic field
Magnetic field
-
 Diachronous
Diachronous
-
 Trachyte
Trachyte
-
 Luyten, Willem
Luyten, Willem
-
 Allosteric effector
Allosteric effector
-
 Key
Key
-
 Transit
Transit
-
 Ischaemic
Ischaemic
-
 Magnetosphere
Magnetosphere
-
 Arable land
Arable land
-
 Migration
Migration
-
 Areole
Areole
-
 Michelson interferometer
Michelson interferometer
-
 Bacillary
Bacillary
-
 Endometriosis
Endometriosis
-
 Caulinary
Caulinary
-
 Retrovirus
Retrovirus
-
 Ectropion
Ectropion
-
 Cave pearls
Cave pearls
-
 P80
P80
-
 Balai
Balai
-
 Asymptomatic
Asymptomatic
-
 Vertebrate
Vertebrate
-
 Hamilton equations
Hamilton equations
-
 Shank feathers
Shank feathers
-
 Rectrice
Rectrice
-
 Gravitational power plants
Gravitational power plants
-
 Polyunsaturated fatty acid
Polyunsaturated fatty acid
-
 Parsnip
Parsnip
Supercooling
In supercooling, matter is in a metastable state in which a liquid of which the temperature is below the freezing point remains in the liquid state instead of solidifying.
The cause of supercooling
This state is due to very slow reaction kinetics and a need at one point for energy in order to change state. However, a small shock (providing energy) or an impurity can cause the matter to change abruptly into whatever stable form is dictated by the pressure and temperature.
 A solidification nucleus in a metastable liquid. © Masakazu "Matto" Matsumoto CC by 2.0
A solidification nucleus in a metastable liquid. © Masakazu "Matto" Matsumoto CC by 2.0
Latest
Fill out my online form.



