-
 Focus
Focus
-
 Perseids
Perseids
-
 Truncation
Truncation
-
 bps bits per second
bps bits per second
-
 Dysaesthesia
Dysaesthesia
-
 Call waiting indication
Call waiting indication
-
 Parsec
Parsec
-
 Archaea
Archaea
-
 Channel D
Channel D
-
 Avascular
Avascular
-
 Autumn equinox
Autumn equinox
-
 Retified wood
Retified wood
-
 Adenocarcinoma
Adenocarcinoma
-
 Common broom
Common broom
-
 Cervix
Cervix
-
 Flood plain
Flood plain
-
 Trackpad
Trackpad
-
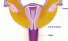 Cervix
Cervix
-
 Focal epilepsy
Focal epilepsy
-
 Salix viminalis
Salix viminalis
-
 Shareware
Shareware
-
 Magnesium
Magnesium
-
 Gallbladder
Gallbladder
-
 Semi-conductor
Semi-conductor
-
 Erenna
Erenna
-
 Pegmatite
Pegmatite
-
 Amalgam
Amalgam
-
 Maculopapular rash
Maculopapular rash
-
 Quantum electrodynamics
Quantum electrodynamics
-
 Gyrus
Gyrus
Fluorine
Fluorine is a chemical element (symbol F), of atomic number 9, atomic mass 18.99, the first element in the halogen group. A pale yellow gas of density 1.696 g/litre (0°C, 1 atm), liquefaction point -188.1°C, freezing point -212.62°C.
Fluorine is prepared by the electrolysis of cryolite or from fluorite (calcium fluoride). Fluorine is a highly reactive oxidiser and is therefore highly toxic. It is used as a fuel or propellant, eventually mixed with oxygen (in a fluorine-hydrogen mixture) as catalyst.
Fluorine is a component of fluorapatite, present in bones, in hydrofluoric acid and fluorinated derivatives such as the (chloro)- fluorocarbons, known commercially as freons, fluorinated polymers such as teflon (PTFE, poly tetrafluoroethylene) and vinylidene polyfluoride (see vinyl).
Latest
Fill out my online form.



