-
 Sub-addressing
Sub-addressing
-
 Oncomodulin
Oncomodulin
-
 Mariner 10
Mariner 10
-
 Electron
Electron
-
 Adduction
Adduction
-
 Protease inhibitor
Protease inhibitor
-
 Specific electricity
Specific electricity
-
 Irregular galaxy
Irregular galaxy
-
 Pallas
Pallas
-
 Meridian of a celestial body
Meridian of a celestial body
-
 Ammeter
Ammeter
-
 Atomic number
Atomic number
-
 Prothorax
Prothorax
-
 Salinity
Salinity
-
 Neurotransmitter
Neurotransmitter
-
 Nebka
Nebka
-
 Elective surgery
Elective surgery
-
 Tholin
Tholin
-
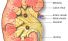 Kidney
Kidney
-
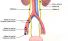 Ureter
Ureter
-
 Stockholm Conference
Stockholm Conference
-
 Genetic polymorphism
Genetic polymorphism
-
 Horizontal coordinates of a direction
Horizontal coordinates of a direction
-
 Final energy
Final energy
-
 OCO
OCO
-
 PRAM
PRAM
-
 Supergiant
Supergiant
-
 Leonids
Leonids
-
 Fibrinolysis
Fibrinolysis
-
 Soft acid
Soft acid
Acid
There are several definitions of an acid (Arrhenius, Brönsted etc.) but the Lewis definition is the widest. This states that an acid is an electron pair acceptor (and hence possesses an empty orbital).
Conjugate acid and base
For every acid there is a conjugate base and vice versa. A base is therefore an electron pair donor. The strengths of an acid and its conjugate base are related. If an acid is strong, its conjugate base is weak, and reciprocally.
General facts about acids
An acid will colour litmus paper red and have a pH lower than 7.
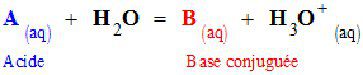
General scheme for the reaction between and acid (A) and water. The chemical equilibrium in the reaction involves the conjugate base (B) of the acid. © DR
Latest
Fill out my online form.



