-
 Channel
Channel
-
 Solar system
Solar system
-
 Epiphysis
Epiphysis
-
 Clone
Clone
-
 Plaster
Plaster
-
 Prostrate shrub
Prostrate shrub
-
 Internal combustion hydrogen engine
Internal combustion hydrogen engine
-
 Commissure
Commissure
-
 Polystyrene
Polystyrene
-
 Planèze
Planèze
-
 ATM
ATM
-
 Asynchronous
Asynchronous
-
 Antibiotic for acne
Antibiotic for acne
-
 Exhalation
Exhalation
-
 Atomic force microscope
Atomic force microscope
-
 Bamako Convention
Bamako Convention
-
 Adware
Adware
-
 Frenulum
Frenulum
-
 Febrile
Febrile
-
 Polyp
Polyp
-
 Proton
Proton
-
 Positron
Positron
-
 Crinoids
Crinoids
-
 Coma
Coma
-
 Eccentricity
Eccentricity
-
 Aldehyde
Aldehyde
-
 Bose-Einstein Condensate
Bose-Einstein Condensate
-
 Rostrum
Rostrum
-
 Shuttering blocks
Shuttering blocks
-
 Chromatid
Chromatid
Protease
A protease (or peptidase) is an enzyme which cleaves protein peptide bonds.
Structure of a protease
Proteases are different and have very different biological roles. Their structures are therefore also very different. However, like all enzymes, they have an active site, which is responsible for their hydrolytic activity on peptide bonds, and a substrate recognition site so that they only act on the target protein.
Function of a protease
These separate two amino acids by breaking the peptide bonds between a carbon atom and a nitrogen atom in the proteins.
The proteases have hydrolase activity as they use a molecule of water to achieve this break. An oxygen and a hydrogen atom from the water molecule make up the carboxyl group on the one hand and on the other, the remaining hydrogen atom is given to the amine group.
Exopeptidases,
- which release the first or last amino acid from the protein ;
- can be distinguished from the endoproteases which cleave the peptide bonds in the middle of proteins.
Proteases are found in digestive juices (such as trypsin or chymotrypsin) but are also involved in cellular processes such as in protein maturation.
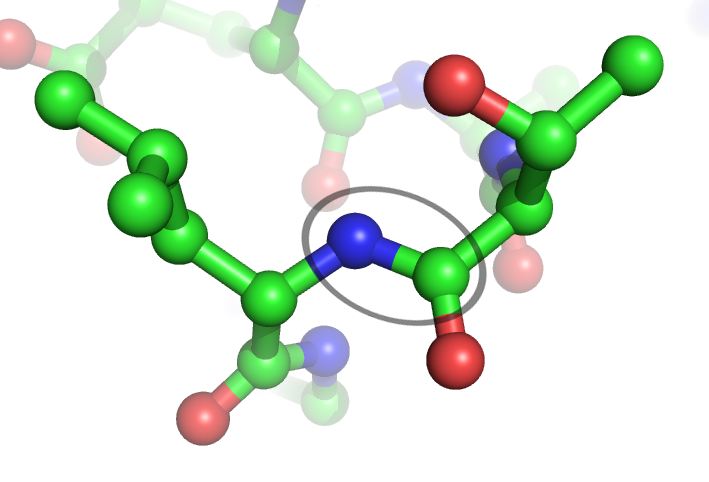 Peptide bonds (between a nitrogen in blue and a carbon in green) are hydrolysed by proteases. © Webridge, Wikimedia, CC by-sa 3.0
Peptide bonds (between a nitrogen in blue and a carbon in green) are hydrolysed by proteases. © Webridge, Wikimedia, CC by-sa 3.0
Latest
Fill out my online form.



