-
 Itching
Itching
-
 Breccia
Breccia
-
 Electromyogram
Electromyogram
-
 Nitrogen vehicle
Nitrogen vehicle
-
 Sponge
Sponge
-
 Sea ice - First year thin
Sea ice - First year thin
-
 Teasel
Teasel
-
 Common ash
Common ash
-
 Macromedia FLASH
Macromedia FLASH
-
 Opt-out
Opt-out
-
 Smart Meter
Smart Meter
-
 Squark
Squark
-
 Hornfel
Hornfel
-
 Leachate
Leachate
-
 StarCraft
StarCraft
-
 Nucleus accumbens
Nucleus accumbens
-
 Facilities management
Facilities management
-
 Pyrrolysine
Pyrrolysine
-
 Pentose
Pentose
-
 Azoospermia
Azoospermia
-
 Feldspars
Feldspars
-
 Equalisation
Equalisation
-
 Reproductive success
Reproductive success
-
 Nitrosamine
Nitrosamine
-
 Coprocessor
Coprocessor
-
 Catechol-O-methyltransferase inhibitor
Catechol-O-methyltransferase inhibitor
-
 Switch
Switch
-
 In vitro fertilisation
In vitro fertilisation
-
 Angiogenesis
Angiogenesis
-
 Small intestine
Small intestine
Lactate
Lactate is a biological molecule which is the ionised form of lactic acid.
Structure of lactate
The chemical structure of lactate is CH3CH-OH-COO-. Lactate has two optical isomers (L or D), but it is the L-lactate form which is produced and metabolised in the body.
Function of lactate
Lactate is the final product of anaerobic glycolysis (fermentation). If glucose consumption exceeds oxygen intake, i.e. during an intense effort, the pyruvate obtained from glycolysis is converted into lactate.
Lactate is taken up in the blood by various human organs or cells. It is used in the liver to produce glucose through gluconeogenesis. It is oxidised in the heart into carbon dioxide (CO2) releasing energy at the same time.
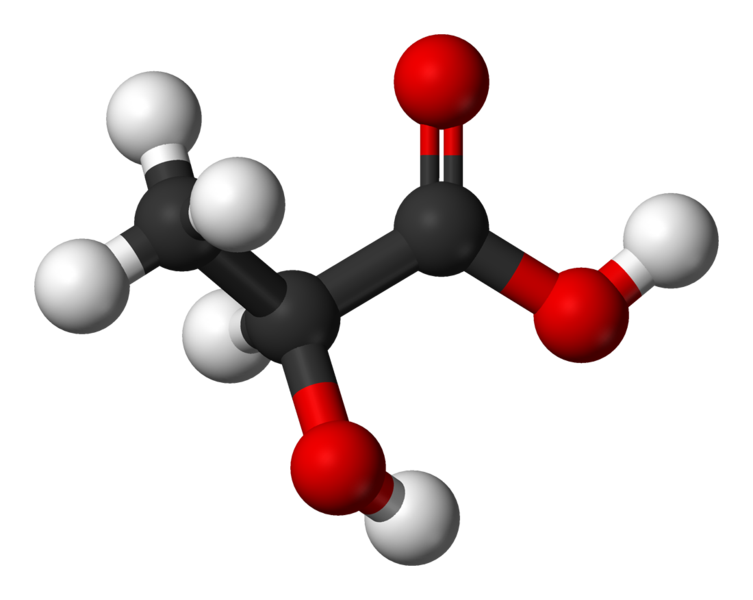 Lactate is the ionic form of lactic acid. © Benjah-bmm27, Wikimedia, public domain
Lactate is the ionic form of lactic acid. © Benjah-bmm27, Wikimedia, public domain
Latest
Fill out my online form.



