-
 Transgenesis
Transgenesis
-
 Wind turbine
Wind turbine
-
 Aspartic acid
Aspartic acid
-
 Smart phone
Smart phone
-
 Aplysia
Aplysia
-
 Common blue
Common blue
-
 Phosphoryl group
Phosphoryl group
-
 Waste sorting
Waste sorting
-
 Mobile blogging
Mobile blogging
-
 Pliocene
Pliocene
-
 Homologous chromosomes
Homologous chromosomes
-
 Monzonite
Monzonite
-
 Twin
Twin
-
 Ginger
Ginger
-
 Onychomycosis
Onychomycosis
-
 Aldolase
Aldolase
-
 ADPCM
ADPCM
-
 Domain name
Domain name
-
 LOX
LOX
-
 Cancer
Cancer
-
 MHC
MHC
-
 Conjecture
Conjecture
-
 Biomass
Biomass
-
 Ecological corridor
Ecological corridor
-
 M71
M71
-
 Alum
Alum
-
 Pentose
Pentose
-
 Nuptial plumage
Nuptial plumage
-
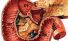 Duodenum
Duodenum
-
 Hormone therapy
Hormone therapy
Carbon monoxide
Carbon monoxide is an oxide of carbon. Its chemical formula is CO so it contains one oxygen atom and one carbon atom .
Carbon monoxide poisoning
Carbon monoxide (CO) is produced by the incomplete combustion of organic fuels (wood, butane, charcoal, petrol, diesel, natural gas, petroleum, and propane.. and is used in heaters to produce hot water or to run motors in electricity generators, for example). It is a highly toxic gas even in small amounts.
Victims of acute poisoning need immediate intensive hospital care with respiratory assistance and oxygenation in a hyperbaric chamber. These accidents can leave lifelong complications.
Concentrations of only 500 ppm (parts per million) of carbon monoxide in respired air cause severe headaches, dizziness and drowsiness indicating the onset of poisoning. Muscle failure and gradual paralysis develop when a person is exposed to concentrations of 2000 ppm, and these are followed by coma without medical assistance. Death also occurs rapidly after exposure to doses of 5000 ppm for only a few minutes. Death occurs when 66% of haemoglobin has been converted into carboxyhaemoglobin.
Studies have shown that blood in the general population in large cities contains 1 to 2% carboxyhaemoglobin. Concentrations in smokers of 4 to 10% make the heart work more in order to oxygenate the body.

The chemical formula of carbon monoxide and interatomic distance. © DR
Latest
Fill out my online form.



